In Rutherford's experiment, some of the alpha particles were deflected indicating the model needed a dense core as a nucleus. In Thomson's plum pudding model, the atom was thought to consist of protons scattered throughout a sphere of negative charge. Describe how Rutherford's gold foil experiment disproved Thomson's model. 3 Oct.Transcribed image text: After he conducted cathode ray tube experiments proving the existence of negatively charged particles we now call electrons, Thomson proposed a model of the atom called the plum pudding model. “The Experimental Discovery of Electron-Thomson Discover.” Think Quest. “J.J Thomson’s Cathode Ray Experiment.”. Style="padding:4px border:1px solid #ddd"> Title="Wordle: Cathode Ray Experiment"> src="" The significance of the mass-to-charge ratio is that two particles with the same mass-to-charge ratio travel in the same pathway in a vacuum when focused on the constant electric and magnetic fields. 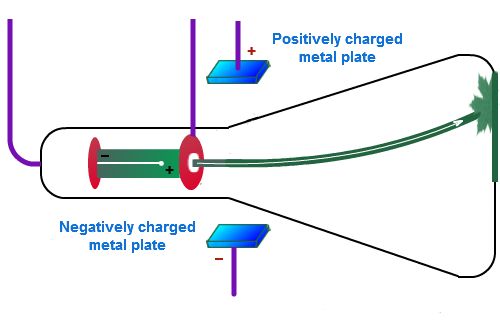
Ernest Rutherford later proved this model incorrect. Thomson imagined the atom as being made up of these corpuscles swarming in an area of positive charge. The “corpuscles” atoms were divisible."^1 (supposed to be a footnote- “J.J Thomson’s Cathode Ray Experiment.”. "He called these particles "corpuscles," and suggested that they might make up all of the matter in atoms. Since these puzzling rays of particles were a lot smaller than atoms Thomson concluded that they were microscopic pieces of atoms. He concluded that the negative cathode ray particles were a thousand times tinier than an atom.Īlong these lines he proved the existence of subatomic particles. Thomson used a charge-to-mass ratio because he knew the weight of the cathode ray tube, the heat, the electrical current, and how much heat had been added from the electrons firing. 
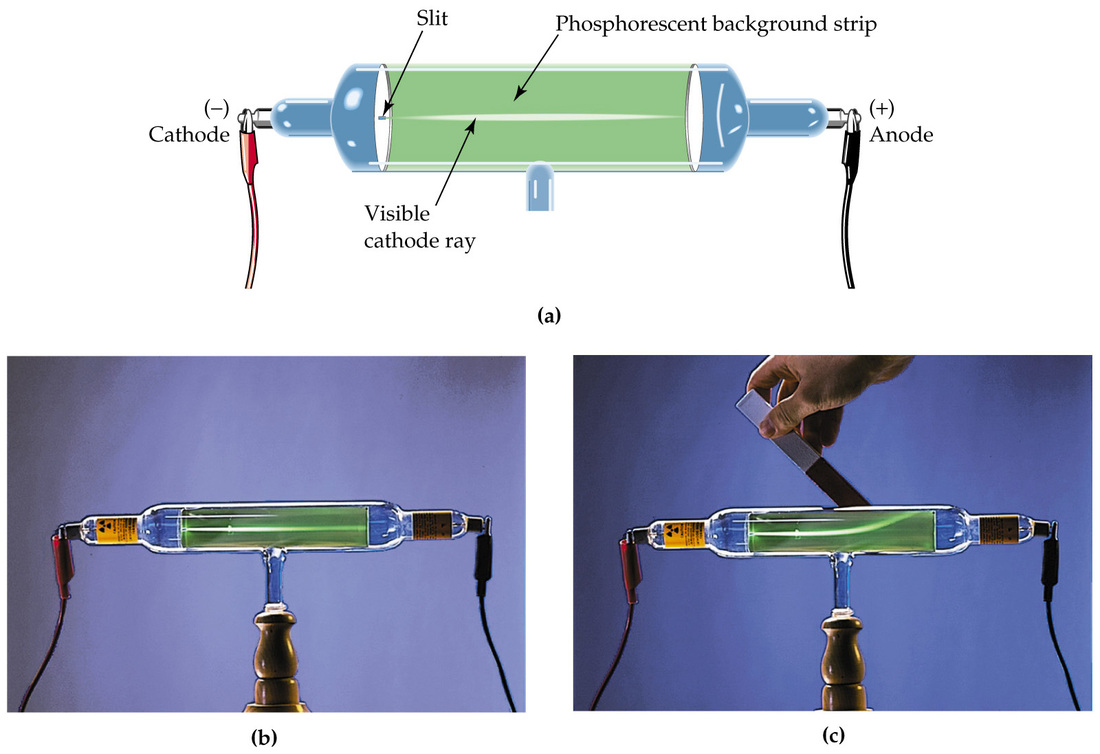
He concluded that cathode rays had a negative charge.Īlthough Thomson had performed all these experiments he was still curious to know what was the size of the cathode rays. The rays were deflected by the electric charge. He would observe this by looking at the light deflection pattern of the cathode ray He knew that electrical charges repelled on another, which allowed him to see whether the charge was a positive or negative. This vacuum had two electrical pates halfway down the tube, one positively charged and one negatively charged. Thomson made a cathode ray with an almost perfect vacuum. His previous experiments did not prove this but he wanted to keep trying because he believed his previous experiment had been flawed because of the traced amounts of gas and a bad vacuum. Thomson’s second experiment was to prove that the rays carried a negative charge. Since the electrometers recorded no activity and so the charge had been bent away by the magnet. He found his conclusion by applying a magnetic field across the tube. Thomson concluded that the negative charge was inseparable from the rays. Therefore, the electrometer registered little charge. Thomson found that if the rays were magnetically bent and they could not enter the slit. This cylinder had two slits in it, leading to electrometers, which could measure small electric charges. To observe this he built a cathode ray tube with a metal cylinder on the end. Thomson’s first experiment was to investigate whether or not the negative charge could be separated from the cathode rays by magnetism. He experimented with currents of electricity inside empty glass tubes. Thomson started the Cathode Ray experiment in the 1894 (19th century) at the Cavendish Laboratory at Cambridge University. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
